Syracuse Views Summer 2024
We want to know how you experience Syracuse University. Take a photo and share it with us. We select photos from a variety of sources. Submit photos of your University experience by filling out a submission form or sending it directly…

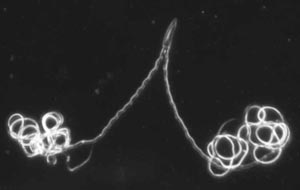 Analyses of the evolutionary relationships among diving beetles reveal that sperm form appears to follow function dictated by female reproductive organs.
Analyses of the evolutionary relationships among diving beetles reveal that sperm form appears to follow function dictated by female reproductive organs.